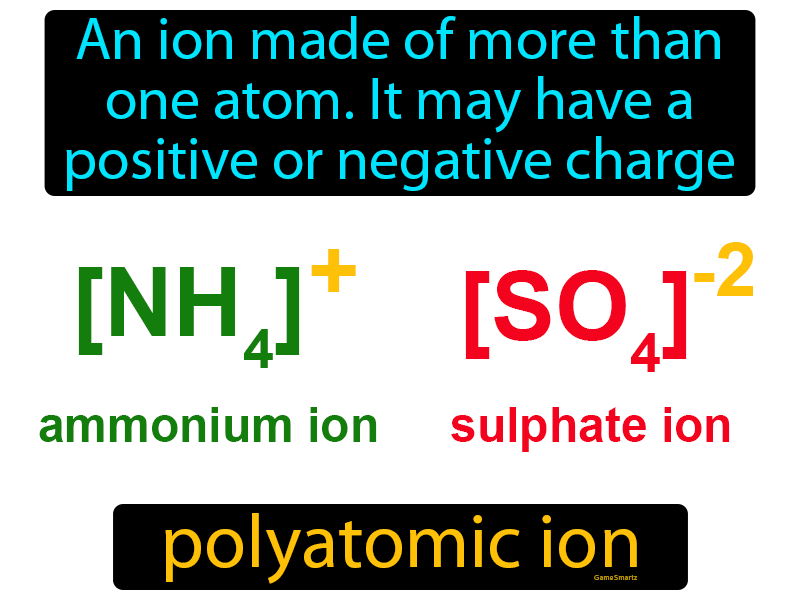
In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Covalent compounds are formed when two nonmetals react with. 
Oxidation and reduction properly refer to a change in oxidation state - the actual transfer of electrons may never occur. Ionic compounds are (usually) formed when a metal reacts with a nonmetal (or a polyatomic ion). When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Compounds formed from such ion combinations are called polyatomic ionic compounds, but the polyatomic ion behaves as a single unit. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. Polyatomic ions are covalently bonded groups of atoms with a positive or negative charge caused by the formation of an ionic bond with another ion. Redox reactions include all chemical reactions in which atoms have their oxidation state changed in general, redox reactions involve the transfer of electrons between species.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
